Viral Inactivation Market by Type, Application and Region - Global Market Analysis and Forecast (2020 - 2025)
The global viral inactivation market is to increase from US$ XX.X million in 2020 to US$ XX.X million by 2025. The market is estimated to grow with a compound annual growth rate (CAGR) of XX.X% (2020-2025). Some of the prominent players in the market are Charles River Laboratories International, Clean Cells, Danaher, Merck, Parker Hannifin, Rad Source Technologies, SGS, Sartorius, Texcell, Viral Inactivated Plasma Systems, Wuxi Pharmatech (Cayman). The research report on the global viral inactivation market provides extensive competition analysis and competitive conditions. The report includes information on significant products, players, challenges and developments, and other information specific to the market.
The global economy is highly affected by the COVID-19. Various sectors in the economy are much affected by this pandemic. The global economy will decline because of the loss of trillions of dollars. The growing extension and imposition of lockdown in various countries directly affect the economy all over the world. The report consists of a chapter that provides a detailed study of the impact of COVID-19 on the viral inactivation market. This report is helpful for business and industry practitioners. The study specifically intended to assist in explaining, directing, and understanding the potential of the viral inactivation markets. The study focuses on providing readers with an understanding of developments in the industry. The report also provides detailed insights on market segments, market forecasts, leading players, market drivers, and inhibitors.
Goals and objectives of the global viral inactivation market study
- Define and discuss the global market for viral inactivation.
- Analyze the various segments of the market as well as the market dynamics of viral inactivation.
- Categorize viral inactivation segments with escalating growth potential and value the segments' future market.
- Analyze the key trends related to different segments and countries that help in figuring the viral inactivation market.
- To check region-specific growth and development in the viral inactivation market.
- Analyze the historical and current value of each of these product segments in the end-user and geographic markets.
- Understand the foremost stakeholders of the market and the value of the competitive landscape of the leaders of the market.
- To study the plans, initiatives, and strategies for the development of the market.
The second chapter, market overview, provides an overview of the viral inactivation market. The chapter discusses macroeconomic factors, drivers, restraints, opportunities, challenges, value chain analysis, technology roadmap, and Porter 5 force model analysis.
Global Viral Inactivation Market Segmentation
Chapter 3, segment analysis provides information on the different sub-segments of the market. The chapter provides an in-depth analysis of the market segments, year-on-year growth projections that enable readers to identify potential market growth areas.
- Based on type, the global viral inactivation market is segmenting into kits and reagents, services, viral inactivation systems and accessories.
- Based on application, the viral inactivation market is segmenting into blood and blood products, cellular and gene therapy products, stem cell products, tissue and tissue products, vaccines and therapeutics.
Global Viral Inactivation Market Regional Analysis
Chapter 4, the regional analysis includes an in-depth analysis of the viral inactivation market by region. The chapter includes various regions such as North America, Europe, Asia-Pacific, Middle East & Africa, and Latin America. The report also includes a comprehensive analysis of the market by country. The report covers countries such as the United States, Canada, Germany, France, Italy, Spain, China, India, Japan, the GCC, South Africa, Brazil, Mexico, and the rest of the world. Regional insight helps market competitors to make crucial decisions about their company. Individualized, country-wise, and segment-wise analyses by region allow readers to explore the potential of the market in different geographies. This section is a vital part of the report for each year's growth projections and a global share of value.
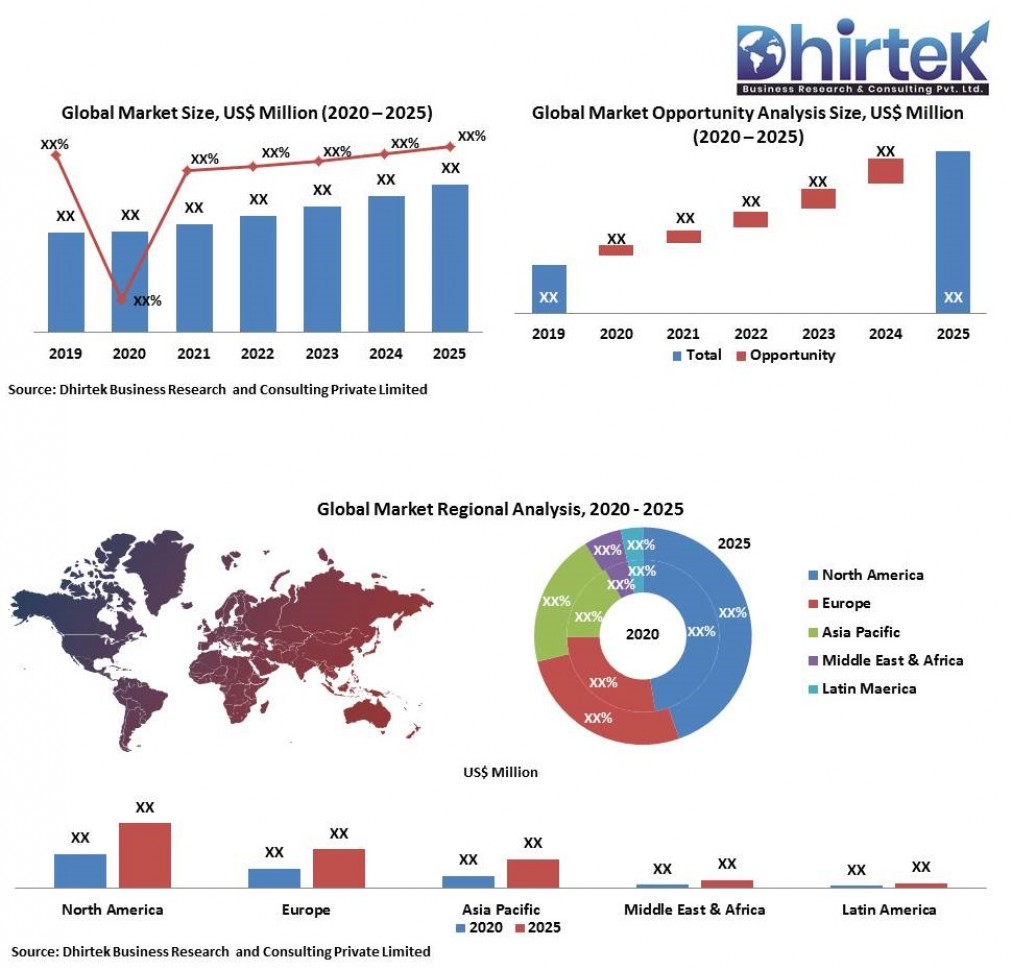
Global Viral Inactivation Market Competitive Analysis
Chapter 5, competitive analysis of the report includes company shares analysis, a list of acquisitions, mergers, collaboration, and the introduction of new products.
Global Viral Inactivation Market Company Profile
Chapter 6, the company profile of the report also covers key industry players working in the viral inactivation market. The report also discusses the business strategies adopted by the players at the global, regional, and country-level. The study also focuses on various businesses or inorganic business development strategies for expanding consumer networks through product formation, organization extension, partnerships, mergers, and acquisitions.
The study offers an exhaustive summary and estimate of the global market. It includes in-depth qualitative and quantitative analyses in the report that align with our customer's goals and objectives. Projections have been confirmed by comprehensive primary and secondary analysis. The study will encourage clients to invest in the market based on the current business situation, trends, and potential developments in the segments. Understanding the requirement of the client, the report delivers specialist insights into the global market, its products, and market patterns.
1. Introduction
2. Market Overview
2.1. Global Viral Inactivation Market Introduction
2.2. Macro- Economic Factor
2.3. Market Determinants
2.3.1. Market Driver
2.3.2. Market Restraints
2.3.3. Market Opportunities
2.3.4. Market Challenges
2.4. Technology/Product Roadmap
2.5. PEST Analysis
2.6. Market Growth Opportunity Analysis
2.7. Impact of Covid-19 on Viral Inactivation Market
3. Market Segmentation
3.1. Global Viral Inactivation Market Analysis (US$ Mn), By Type, 2019 - 2025
3.1.1 Kits and Reagents
3.1.2 Services
3.1.3 Viral Inactivation Systems and Accessories
3.2. Global Viral Inactivation Market Analysis (US$ Mn), By Application, 2019 - 2025
3.2.1 Blood and Blood Products
3.2.2 Cellular and Gene Therapy Products
3.2.3 Stem Cell Products
3.2.4 Tissue and Tissue Products
3.2.5 Vaccines and Therapeutics
4. Regional Analysis
4.1. North America Viral Inactivation Market Analysis (US$ Mn), 2019 - 2025
4.1.1. By Country
4.1.1.1. U.S.
4.1.1.2.Canada
4.1.2.By Type
4.1.3.By Application
4.2.Europe Viral Inactivation Market Analysis (US$ Mn), 2019 - 2025
4.2.1.By Country
4.2.1.1.Germany
4.2.1.2.U.K.
4.2.1.3.France
4.2.1.4.Italy
4.2.1.5.Spain
4.2.1.6.Rest of Europe
4.2.2.By Type
4.2.3.By Application
4.3.Asia Pacific Viral Inactivation Market Analysis (US$ Mn), 2019 - 2025
4.3.1.By Country
4.3.1.1.China
4.3.1.2.Japan
4.3.1.3.India
4.3.1.4.Rest of Asia Pacific
4.3.2.By Type
4.3.3.By Application
4.4.Rest of world Viral Inactivation Market Analysis (US$ Mn), 2019 - 2025
4.4.1. By Region
4.4.1.1. Middle East & Africa
4.4.1.2. Latin America
4.4.2.By Type
4.4.3. By Application
5.Company Profiles
5.1 Charles River Laboratories International
5.2 Clean Cells
5.3 Danaher
5.4 Merck
5.5 Parker Hannifin
5.6 Rad Source Technologies
5.7 SGS
5.8 Sartorius
5.9 Texcell
5.10 Viral Inactivated Plasma Systems
5.11 Wuxi Pharmatech (Cayman)